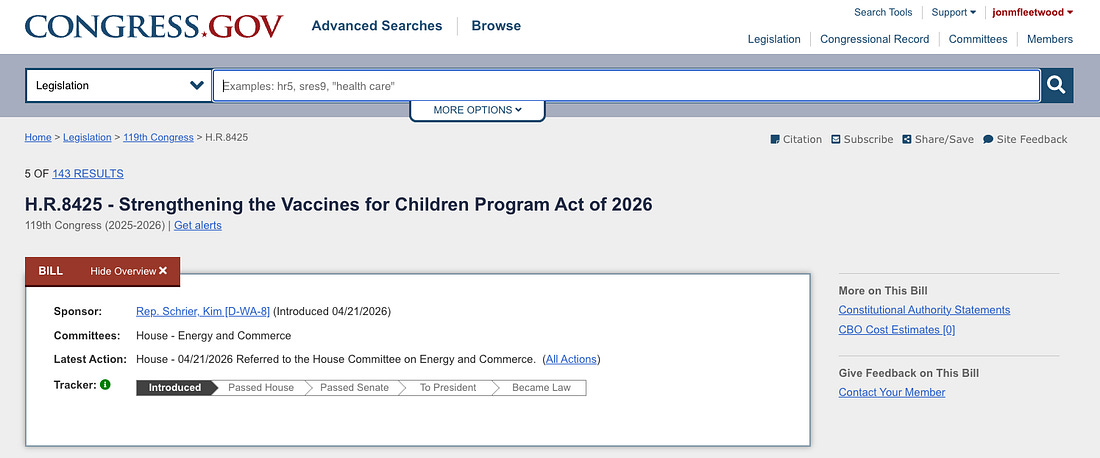
Congress Introduces Billion-Dollar Child Vaccine Control Grid: H.R. 8425
Rep. Schrier’s bill funnels taxpayer cash into Big Pharma, state propaganda, and pediatric surveillance expansion.

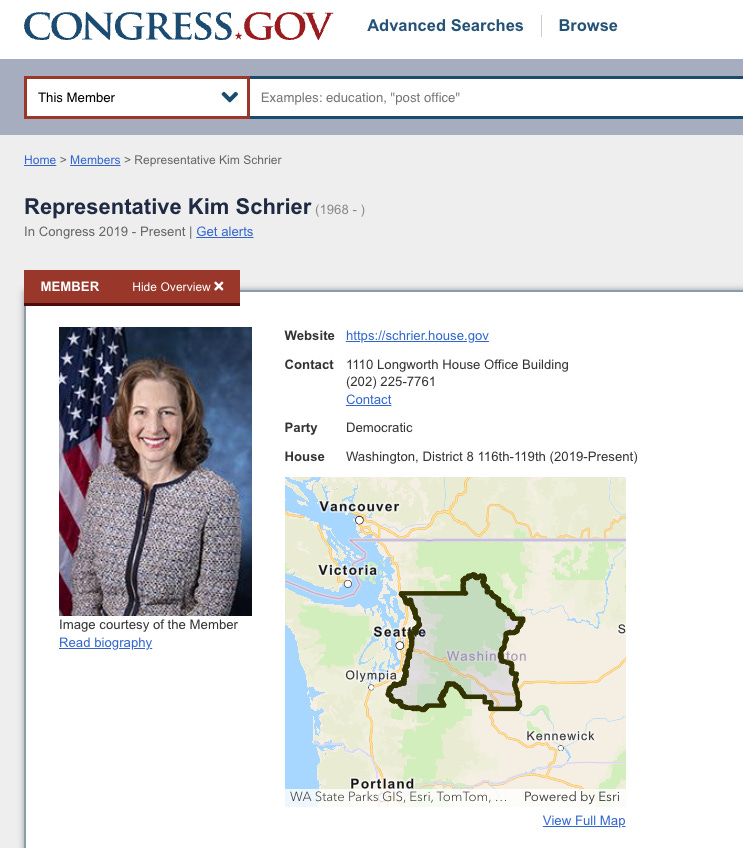
Representative Kim Schrier (D-WA) last week introduced H.R. 8425, the “Strengthening the Vaccines for Children Program Act of 2026,” a sweeping federal bill that could funnel billions in taxpayer dollars into an expanded child vaccine control grid.
The legislation does this by deepening federal vaccine infrastructure, increasing payments to vaccine administrators, financially coercing states into running federally approved vaccine propaganda campaigns, and expanding long-term pediatric surveillance systems.
According to campaign finance watchdog OpenSecrets and the Federal Election Commission, Schrier’s donor base includes entities with potential financial or institutional interests in expanded vaccine systems, including:
- Pfizer
- Abbott Laboratories
- Quest Diagnostics
- Kaiser Permanente
- American Medical Association
Critics view this as a direct conflict of interest, with pharmaceutical and medical industry donors financially backing a lawmaker whose bill could materially benefit the very corporations and healthcare systems funding her political career.
Rep. Schrier introduced H.R. 8425 alongside original cosponsor Rep. John Joyce (R-PA).
Additional cosponsors include Rep. Suzan DelBene (D-WA), Rep. Josh Riley (D-NY), Rep. Joseph Morelle (D-NY), Rep. Mike Quigley (D-IL), Rep. Henry Johnson (D-GA), and Rep. Grace Meng (D-NY).
You can contact the representatives listed above by clicking through the links in their names.
You can find your representative here and let them know how you would like them to vote on the bill.
H.R. 8425 was immediately referred to the House Committee on Energy and Commerce, where it currently remains in committee after introduction.

Federal Government Uses Medicaid Billions to Pressure States Into Running Vaccine Propaganda
Beginning January 1, 2027, H.R. 8425 offers states a 1% increase in Federal Medical Assistance Percentage (FMAP)—a potentially multi-billion-dollar taxpayer-funded incentive—but only if they comply with federal vaccine messaging mandates.
The bill explicitly states:
“Federal medical assistance percentage determined for each State… under section 1905(b) of the Social Security Act (42 U.S.C. 1396d(b)) shall be increased by 1 percentage point.”
“A State… may not receive the increase… if such State does not ensure culturally competent and effective messages for vaccination outreach to child populations…”

Required messaging includes promotion of:
“advancements in research and vaccine development that have saved millions…”
“the dangers of not being vaccinated…”
“vaccine safety…”
This creates direct federal financial leverage to transform state health departments into taxpayer-funded child vaccine propaganda systems.
Bill Expands Federal Child Vaccine Pipeline Into Millions More Children
The bill broadens federal vaccine system reach by expanding eligibility:
“A child who is enrolled for child health assistance under a State child health plan approved under title XXI.”
This automatically folds CHIP-enrolled children deeper into federally subsidized vaccine programs, expanding the national pediatric vaccine apparatus.
According to the Centers for Medicare & Medicaid Services (CMS), 7,243,961 children were enrolled in CHIP as of December 2025.
Providers Paid Premium Rates to Push Vaccines—Even When Parents Decline
H.R. 8425 guarantees:
“payment for vaccine administration and counseling services… at a rate not less than 100 percent…”
And providers may bill:
“regardless of whether such vaccine is actually administered”
This means taxpayer dollars can directly reward doctors and healthcare systems for vaccine pressure campaigns even when families refuse injections.
Combination Vaccines Become Bigger Big Pharma Revenue Engines
The bill authorizes:
“a separate charge for the administration of and counseling for each component of such vaccine”
This creates stronger reimbursement incentives for expanded multi-component vaccine schedules, potentially increasing pharmaceutical and provider profits.
Pediatric Surveillance Grid Deepened
The legislation authorizes broader access to:
“data, data sets, monitoring systems, delivery systems, and other protected health information…”
CDC must also publicly track:
“vaccination rates… disaggregated by region, age, sex, race, ethnicity…”
This would significantly expand the federal government’s pediatric surveillance grid by increasing institutional access to protected child health data, strengthening vaccine uptake monitoring systems, and building more powerful demographic tracking infrastructure capable of identifying, targeting, and pressuring under-vaccinated populations with greater precision.
Bottom Line
H.R. 8425 is a major federal expansion of a billion-dollar child vaccine control grid that could:
- Funnel taxpayer money into pharmaceutical and provider systems
- Financially coerce states into vaccine propaganda compliance
- Reward providers for vaccine pressure
- Expand federally managed child vaccine pipelines
- Build stronger surveillance and demographic compliance tracking systems
For critics focused on medical freedom, parental rights, and government overreach, the bill represents a substantial escalation in the merger of federal power, pharmaceutical profit, and public health surveillance—building the infrastructure today for larger future child vaccine campaigns, broader compliance pressure, and deeper institutional control tomorrow.

























Recent Comments